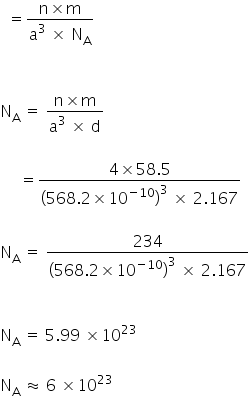Avogadro constant: Numerical value: 6.022 140 76 x 10 23 mol-1: Standard uncertainty (exact) Relative standard uncertainty (exact) Concise form 6.022 140 76 x 10 23 mol-1. Avogadro’s number, number of units in one mole of any substance (defined as its molecular weight in grams), equal to 6.02214076 × 10 23. The units may be electrons, atoms, ions, or molecules, depending on the nature of the substance and the character of the reaction (if any). See also Avogadro’s law. Avogadro's number is named in honor of the Italian scientist Amedeo Avogadro. Mc marine gps gmr900 driver for mac. Although Avogadro proposed that the volume of a gas at a fixed temperature and pressure was proportional to the number of particles it contained, he did not propose the constant. In 1909, French physicist Jean Perrin proposed Avogadro's number. Click here👆to get an answer to your question ️ If NA is Avogadro's number, then number of valence electrons in 4.2 g of nitride ion ( N^3 - ) is.
2.4 $N_A$
C1.6$N_A$
Solution:
$14,g$ of $N^{3-}$ ions have valence electrons $=8N_{A}$
$4.2,g$ of $N^{3-}$ ions have valence electrons
$=frac{8N_{A}times 4.2}{14}=2.4,N_{A}$
1. A metal M (specific heat 0.16) forms a metal chloride with = 65% chlorine present in it. The formula of the metal chloride will be
2. Mass % of carbon in ethanol is
3. Which element has the highest first ionization potential?
4. Which statement(s) is (are) false for the periodic classification of elements?
5. The electronegativities of N, C, Si and P are in the order
Na Avogadrov Broj
6. The effective nuclear charge of an element with three valence electrons is 2.60. What is the atomic number of the element?
7. Identify the element that forms amphoteric oxide.
8. Which of the set of oxides are arranged in the proper order of basic, amphoteric, acidic ?
9. 5 moles of $AB^2$ weigh $125 times 10^{-3}; kg$ and 10 moles of $A_2B_2$ weigh $300 times 10^{-3} ; kg$. The molar mass of $A(M_A)$ and molar mass of $B(M_B) $ in kg mol$^{-1}$ are :
10. Which of the following is an example of Law of multiple proportion?
1. In Wolff‐Kishner reduction, the carbonyl group of aldehydes and ketones is converted into
2. Identify compound X in the following sequence of reactions:

3. Identify a molecule which does not exist.
4. Identify the incorrect match.
Name IUPAC Official Name A Unnilunium i Mendelevium B Unniltrium ii Lawrencium C Unnilhexium iii Seaborgium D Unununnium iv Darmstadtium
| Name | IUPAC Official Name | ||
|---|---|---|---|
| A | Unnilunium | i | Mendelevium |
| B | Unniltrium | ii | Lawrencium |
| C | Unnilhexium | iii | Seaborgium |
| D | Unununnium | iv | Darmstadtium |
5. Reaction between acetone and methyl magnesium chloride followed by hydrolysis will give :
6. Identify the correct statements from the following:
(a) $CO_2(g)$ is used as refrigerant for ice-cream and frozen food.
(b) The structure of $C_{60}$ contains twelve six carbon rings and twenty five carbon rings.
(c) $ZSM-5$, a type of zeolite, is used to convert alcohols into gasoline.
(d) $CO$ is colorless and odourless gas.
7. Which of the following set of molecules will have zero dipole moment ?
8. On electrolysis of dil.sulphuric acid using Platinum (Pt) electrode, the product obtained at anode will be:
9. An element has a body centered cubic (bcc) structure with a cell edge of 288 pm. The atomic radiusis:
10. Find out the solubility of $Ni(OH)_2$ in 0.1 M NaOH. Given that the ionic product of $Ni(OH)_2$ is $2 times 10^{-15}$
Contrary to the beliefs of generations of chemistry students, Avogadro’s number—the number of particles in a unit known as a mole—was not discovered by Amadeo Avogadro (1776-1856). Avogadro was a lawyer who became interested in mathematics and physics, and in 1820 he became the first professor of physics in Italy. Avogadro is most famous for his hypothesis that equal volumes of different gases at the same temperature and pressure contain the same number of particles.
Na Avogadro's Constant
The first person to estimate the actual number of particles in a given amount of a substance was Josef Loschmidt, an Austrian high school teacher who later became a professor at the University of Vienna. In 1865 Loschmidt used kinetic molecular theory to estimate the number of particles in one cubic centimeter of gas at standard conditions. This quantity is now known as the Loschmidt constant, and the accepted value of this constant is 2.6867773 x 1025 m-3. Hy ad001 user manual.
The term “Avogadro’s number” was first used by French physicist Jean Baptiste Perrin. In 1909 Perrin reported an estimate of Avogadro’s number based on his work on Brownian motion—the random movement of microscopic particles suspended in a liquid or gas. In the years since then, a variety of techniques have been used to estimate the magnitude of this fundamental constant.
Na Avogadrova Konstanta
Accurate determinations of Avogadro’s number require the measurement of a single quantity on both the atomic and macroscopic scales using the same unit of measurement. This became possible for the first time when American physicist Robert Millikan measured the charge on an electron. The charge on a mole of electrons had been known for some time and is the constant called the Faraday. The best estimate of the value of a Faraday, according to the National Institute of Standards and Technology (NIST), is 96,485.3383 coulombs per mole of electrons. The best estimate of the charge on an electron based on modern experiments is 1.60217653 x 10-19 coulombs per electron. Photoninja. If you divide the charge on a mole of electrons by the charge on a single electron you obtain a value of Avogadro’s number of 6.02214154 x 1023 particles per mole.
Another approach to determining Avogadro’s number starts with careful measurements of the density of an ultrapure sample of a material on the macroscopic scale. The density of this material on the atomic scale is then measured by using x-ray diffraction techniques to determine the number of atoms per unit cell in the crystal and the distance between the equivalent points that define the unit cell (see Physical Review Letters, 1974, 33, 464).